The tools and tests used to diagnose inherited retinal disease (IRD)
June 14, 2019
Diagnosing rare diseases like retinitis pigmentosa (RP), Leber congenital amaurosis (LCA), or choroideremia can be complex. Here are a few of the tools eye care professionals often use along the diagnostic journey.
This article is for informational purposes only and is not meant to serve as medical advice. Please talk to your eye care professional should you have questions about the health of your eyes.
Most diagnostic journeys for those with inherited vision loss start in the same way they do for anyone going to see an eye care professional—with a medical history and vision exam. This baseline exam will measure your visual acuity and ability to distinguish colors, and help identify the presence of cataracts, glaucoma, or a retinal disorder. Should your eye care professional suspect that your vision loss is due to a particular inherited retinal disease (IRD), like retinitis pigmentosa (RP), Leber congenital amaurosis (LCA), choroideremia, or Stargardt disease, you may undergo further testing. Each person’s situation is different, and their eye care professionals will determine which tests to conduct during the diagnostic journey.
Some of these tools and tests are complex, and you may have questions. While you will find plenty of online information about the various tests and procedures, we wanted to break it down in understandable language—life is complicated enough without having to decipher medical jargon.
It’s now possible to identify the genetic mutation(s) in up to 55 to 80 percent of patients with an IRD.1”
First of all, it’s important to delineate between a clinical and a genetic diagnosis. A clinical diagnosis is based on your clinician’s observation of the signs and symptoms you exhibit. Since IRDs are characterized by the loss of photoreceptors and the retinal pigment epithelium (RPE) function2, your vision care team may use several tools or tests to assess the health of these key indicators, identify your disease, and then track its progression.
A genetic diagnosis, which differs from a clinical diagnosis, can only be determined by testing your DNA. Your physician will collect a small sample of a bodily fluid—most commonly blood or saliva—and send the sample off to a genetic testing lab. A much more specific diagnostic method, genetic testing is able to identify the genetic mutation(s) responsible for your IRD. A genetic diagnosis can help your eye care professional provide more tailored disease management and identify potential treatments or clinical trials for you to consider.
Clinical diagnosis
On your road to a clinical diagnosis, these are some of the clinical tests you may encounter:
Comprehensive, dilated eye exam3
This baseline test gives eye care professionals a clear view of the retina, including the central macula region and the periphery, as well as the optic nerve and retinal blood vessels, to help determine overall eye health and identify potential issues. Eye drops dilate the pupils, which provides a better sightline to the back of the eye. Your eye care professional then can use several different types of magnifying glasses to assess the current health of your eye tissue.
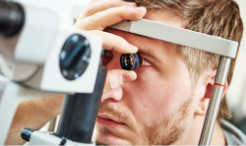
Most diagnostic journeys start with a comprehensive eye exam.
Visual field test3
The visual field test is a common vision test to detect dysfunction in central and peripheral vision. During the test you look straight ahead while a light or images are generated in your side—or peripheral—vision and move toward your center vision. As you indicate the times you see the light or images flash, your eye care professional will map your visual field boundaries. Visual field testing is useful to monitor the progression of disease and document the status of legal blindness.
Retinal imaging4
Digital retinal imaging can detect disease early as well as provide an ongoing, historical record of changes in your eyes. Retinal imaging captures miniscule retinal structures and abnormalities through the use of special cameras. These cameras take photos of your retina to help spot disease characteristics and track progression (see below for more retinal imaging information). The noninvasive imaging of each eye’s fundus and retina, including the central macula region and the periphery, as well as the optic nerve and retinal blood vessels, can also clinically confirm a suspected diagnosis and direct decisions about which genetic tests to order.
Electrophysiological testing includes a number of tests that provide information about a person’s vision beyond the standard clinical eye examination. They are designed to test the ability of your eyes to move visual information from the retina, at the back of the eye, through the visual pathways to the visual center of the brain.
- Full-field electroretinogram (ERG)5
The ERG measures the function of the retina by testing the response of the eyes’ photoreceptors, or rods and cones, as well as several other cells that process these light signals before they are sent to the brain. An electrode is placed on the eye and attached to a wire that will transmit a record of retinal activity. As you look into a large reflective globe, lights are flashed into the eyes. The wave patterns generated by the response of photoreceptors to light can help an eye care professional diagnose an IRD.Note: Young children undergoing an ERG will often be sedated or put under general anesthesia for the procedure.
- Dark adaptometry (DA)5
The DA test assesses the ability of your eyes to adapt to the dark. The test measures the length of time it takes for the retina to regain maximum sensitivity to low amounts of light after it has been exposed to bright light and then returned to darkness. The test can be useful in detecting early cases of IRDs that include nyctalopia, or night blindness, as a symptom.
Genetic diagnosis
While the imaging and tests listed above are still key for a clinical diagnosis, genetic testing is the only way to uncover the genetic mutation(s) responsible for your IRD. Genetic testing has advanced significantly in recent years. Next-generation sequencing (NGS) now uses targeted panels of all known retinal dystrophy genes and makes it possible to identify the genetic mutation(s) in up to 55 to 80 percent of patients with an IRD1. That’s why genetic testing has now become an essential step in the diagnosis of IRDs.
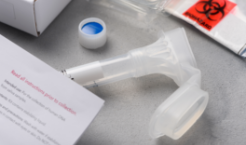
In some cases, a small vial of saliva provides the DNA needed for genetic testing.6
Providing DNA for the test itself is relatively simple. In some cases, your physician will collect a small sample of saliva or blood and send it off to a lab for processing.6 Once results are in, which can take anywhere from a few weeks to months, your doctor will discuss the findings, implications, and next steps with you.
Identifying your genetic mutation(s) can:
- Definitively confirm your clinical diagnosis and detect any associated diseases or conditions
- Offer genetic information for family planning purposes
- Give extended family members important insight about their own health or genetics
- Help tailor disease management plans, including providing information about ongoing or future clinical trials and potential new treatments
Each person’s diagnostic journey is different and depends on several factors—the kind of eye care professional you see, which specific IRD is suspected to be causing the vision loss, and the types of tests that are used. In general, eye care specialists will want to identify which IRD is at play through a clinical diagnosis before calling for genetic testing. Your eye care professional will base that clinical diagnosis on one or more of the tests outlined here, coupled with the retinal imaging mandated by your signs and symptoms. The only way to confirm a clinical diagnosis and identify the mutation(s) responsible for your IRD is through genetic testing.
Options for retinal imaging include:
- Conventional color fundus imaging
Fundus photography captures a photo of the back of the eye with a microscope fitted to a flash-enabled camera. The image visualizes the central and peripheral retina, optic disk, and macula.7
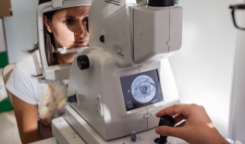
- Scanning laser ophthalmoscopy (SLO)
An infrared beam rapidly scans the retina to detect degenerative changes in photoreceptor and retinal pigment epithelium (RPE) cell layers. The SLO is able to image structures at very high magnification and frame rate, providing high resolution imaging to identify the presence or absence of key retinal structures that are hard to see with ordinary fundus imagery.8
- Optical coherence tomography (OCT)
OCT uses light waves to take a cross-section picture of your retina. It is particularly useful for examining retinal architecture, including the outer retina structure and thickness, and the photoreceptor layer.9

Images of the retina provided by new OCT technology give clinicians a view of the choroid not available with other imaging techniques.9
- Adaptive optics (AO) imaging: The rapid advancements produced as a result of retinal research have resulted in progressively better imaging options like AO retinal imaging.10 This new technology can visualize photoreceptors as well as the retinal RPE, retinal nerve fiber layer, retinal vessel wall, and the lamina cribosa (an important element of the optic nerve architecture).11, 12
- Fundus autofluorescence imaging (FAF): Fundus autofluorescence is an imaging method for mapping changes in the RPE. The technique relies on the fluorescent properties of pigments in the retina to generate images13 and can be used in combination with other imaging techniques.
1. Glockle N, Kohl S, Mohr J, et al. Panel-based next generation sequencing as a reliable and efficient technique to detect mutations in unselected patients with retinal dystrophies. Eur J Hum Genet, 2014;22(1):99-104.
2. Hafler BP. Clinical progress in inherited retinal degenerations: Gene therapy clinical trials and advances in genetic sequencing. Retina. 2017;37(3):417-423. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5465814/
3. Duffy M. The difference between a vision screening and a comprehensive eye examination. VisionAware. https://visionaware.org/screen-magnification-software/your-eye-condition/eye-health/eye-examination/. Accessed May 22, 2019.
4. American Academy of Ophthalmology. Recommendations on clinical assessment of patients with inherited retinal degenerations. https://www.aao.org/clinical-statement/recommendations-on-clinical-assessment-of-patients. Accessed May 22, 2019.
5. University of Iowa Health Care, Carver College of Medicine, Department of Ophthalmology and Visual Sciences. Ophthalmic electrophysiology. https://medicine.uiowa.edu/eye/patient-care/labs-and-screening/ophthalmic-electrophysiology. Accessed May 22, 2019.
6. Genetics Home Reference, US National Library of Medicine, National Institutes of Health. How is genetic testing done? https://medlineplus.gov/genetics/understanding/testing/procedure/. Accessed May 22, 2019.
7. Ophthalmic Photographers’ Society. Fundus Photography Overview. https://www.opsweb.org/page/fundusphotography. Accessed May 22, 2019.
8. American Academy of Ophthalmology. Scanning laser ophthalmoscopy. https://www.aao.org/bcscsnippetdetail.aspx?id=91f993d5-5903-41c2-bc0a-8f19679df403. Accessed May 21, 2019.
9. Adhi M, Duker J. Optical coherence tomography—current and future applications. NIH Public Access, National Institutes of Health. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3758124/pdf/nihms-506055.pdf. Accessed May 23, 2019.
10. Godara P, Dubix A, Roorda A, et al. Adaptive optics retinal imaging: Emerging clinical applications. American Academy of Optometry. 2010; 87(12):930-941.
11. Kozak I. Retinal imaging using adaptive optics technology. Saudi J Ophthalmol. 2014 Apr; 28(2): 117–122. doi: 10.1016/j.sjopt.2014.02.005.
12. Jonas J, Berenshtein E, Holback L. Anatomic relationship between lamina cribrosa, intraocular space, and cerebrospinal fluid space. Investigative Ophthalmology & Visual Science. 2003 December; 44(12):5189-5195.
13. Sepah YJ, Akhtar A, Sadiq MA, et al. Fundus autofluorescence imaging: Fundamentals and clinical relevance. Saudi J Ophthalmol. 2014;28(2):111-116.
Sign up to find out when we add new stories to the site.
